 
In contrast, a nitro substituent decreases the ring's reactivity by roughly a million. For example, a hydroxy or methoxy substituent increases the rate of electrophilic substitution about ten thousand fold, as illustrated by the case of anisole in the virtual demonstration (above). Experiments have shown that substituents on a benzene ring can influence reactivity in a profound manner. The first is the relative reactivity of the compound compared with benzene itself. When substituted benzene compounds undergo electrophilic substitution reactions of the kind discussed above, two related features must be considered: Thus, a sigma bond between sp 2- and sp 3‑carbon is slightly polarized, as follows: Note that one argument sometimes used to explain the ability of alkyl groups to donate electrons inductively to an aromatic ring is that sp 2‑hybridized carbon atoms are more electronegative than sp 3‑hybridized carbon atoms. 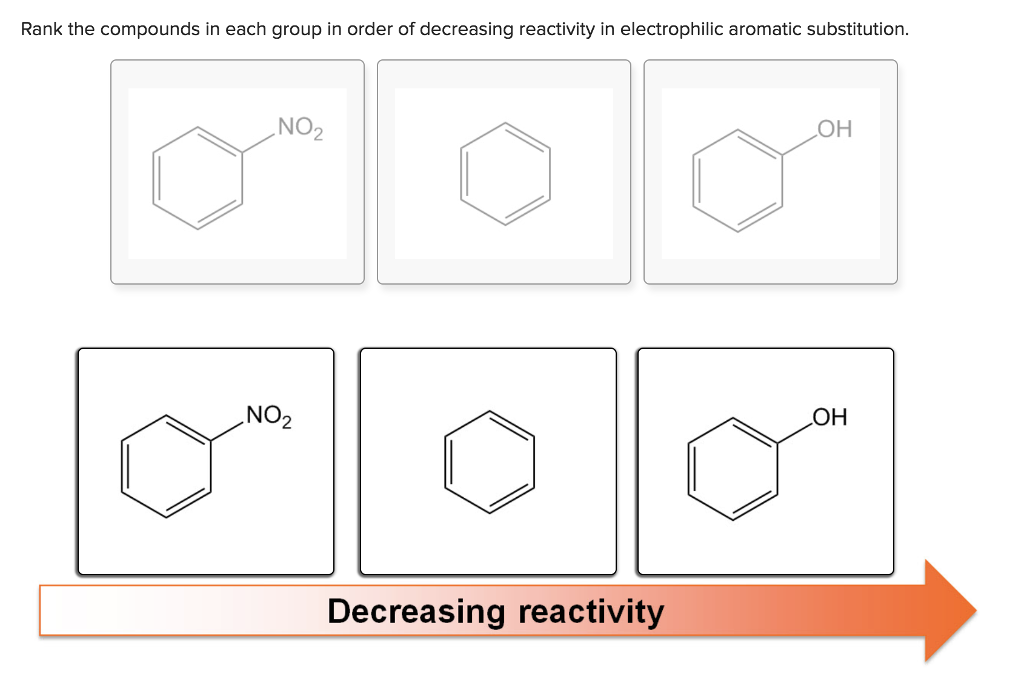
If so, refer to Sections 2.1, 7.9 (paying particular attention to the “Study Notes”) and 14.5. The first step of electrophilic aromatic substitution is usually the rate-determining step. 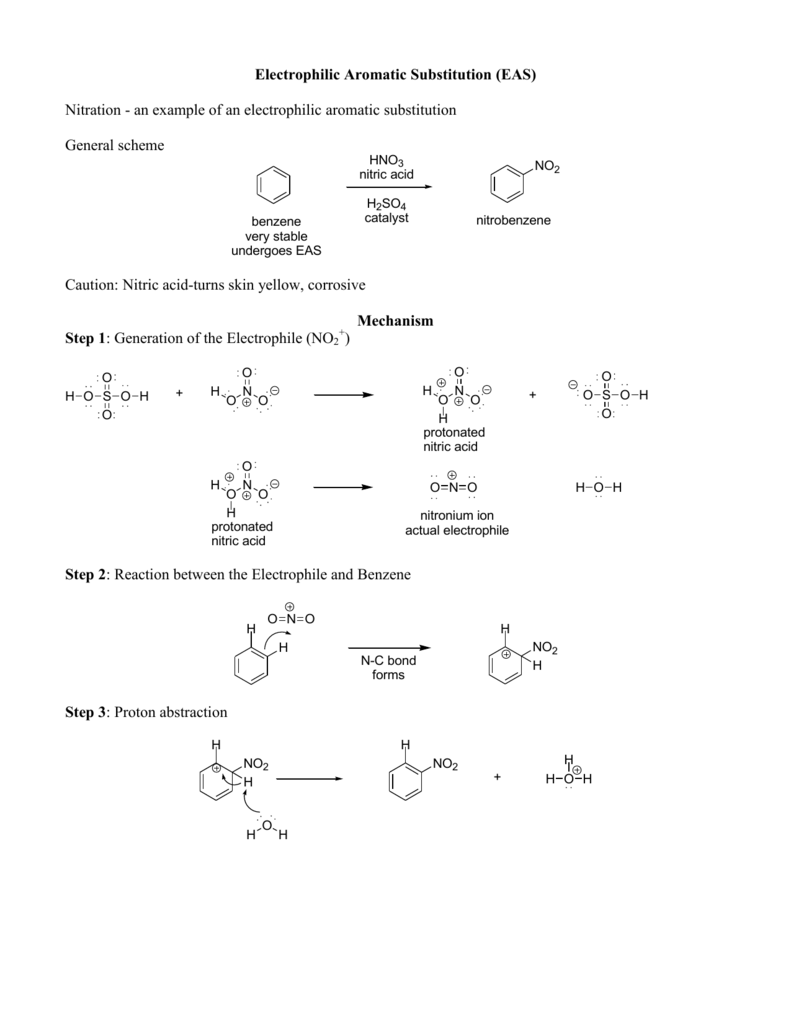
You may wish to review earlier material on to the inductive effect. What you should be doing is trying to understand the factors that determine whether a given substituent will activate or deactivate a benzene ring with respect to electrophilic substitution. The answer is that if you are trying to memorize such things, you are taking the wrong approach to organic chemistry. On reading Objective 2 students may exclaim “How am I ever going to memorize all of this!”-or words to that effect. explain the resonance effects displayed by substituents such as nitro, carbonyl-containing, hydroxy, alkoxy and amino groups during electrophilic aromatic substitution reactions.explain the inductive effects displayed by substituents such as nitro, carboxyl, alkyl and the halogens during electrophilic aromatic substitution reactions.list a given series of aromatic compounds in order of increasing or decreasing reactivity with respect to electrophilic substitution.Arenes contain double bonds just like alkenes but they do not undergo electrophilic addition because these would result to their loss of ring aromaticity. explain, in general terms, the factors that determine whether a given substituent will activate or deactivate an aromatic ring with respect to electrophilic substitution. Aromatic compounds or arenes undergo substitution reactions, in which the aromatic hydrogen is replaced with an electrophile, hence their reactions proceed via electrophilic substitution.list a given series of substituents (selected from those given in Objective 2) in order of increasing or decreasing ability to activate or deactivate an aromatic ring with respect to electrophilic substitution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
